An indication selection resource for longevity companies
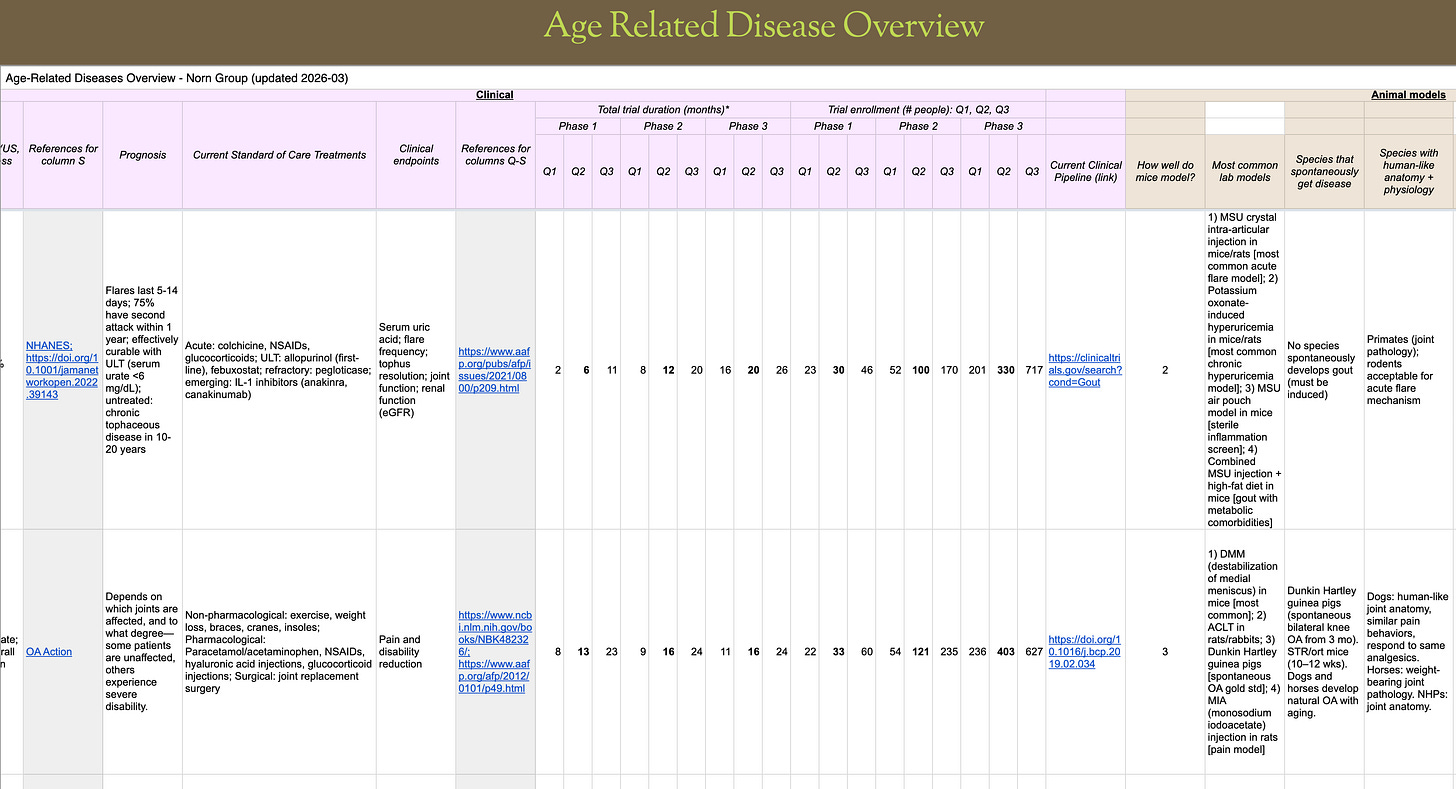
We're sharing a spreadsheet with key data on 47 diseases that aging drugs could potentially treat, to help fresh startups and academic scientists pick viable drug development paths.
Taking a longevity treatment from the lab to a real drug program involves a lot of hard decisions, especially if you’re also forming a company to do it. One of the most critical is deciding what you want your drug to eventually be FDA approved to do, and we’re sharing a new resource to help you do that1.
Why?
Unless your company has access to at least a billion mission-aligned dollars, you will find that targeting ‘longevity’ is not a viable path. There is not yet a reimbursable endpoint for aging, no defined approval criteria, and no previous established commercial model for running an aging trial. We believe this will change (and are actively working to accelerate that with e.g. multi-morbidity trial designs for aging and biomarker validation programs). But until that is done, investors will want to see a path to profit that doesn’t involve figuring out new ways to do (long) clinical trials and new ways to pay for medicines in the US healthcare system2. This means picking a recognized disease where you think your drug will work, with plans to expand to additional diseases and/or preventative treatments eventually.
99% of all longevity therapeutic companies end up on this path, even when the founders want to cure aging. BioAge did, NewLimit did, etc. Some do so right away, others push the longevity goal until they’re no longer able to raise funding for that, then adjust plans. We at Norn Group think about the big picture of producing longevity, and care about companies succeeding. So a while back we decided to collect all the most important information for indication selection for every age-related disease we could think of, and serve this up to the community to save everyone else from having to do the same. We’ve now updated the table to add additional diseases and represent current standard of care.
How to use this resource
The spreadsheet covers 47 age-related indications across various organ systems. For each indication, we have included a brief overview of disease mechanism and links to mechanisms of aging, data on human incidence and patient population, current treatment options and clinical endpoints/measurables, relevant animal models of the disease and their relevance to humans, size and duration of clinical trials, as well as market size. This should give you most of the information needed to consider the best fit for your therapeutic candidates. We suggest paying extra attention to the following:
Where does your biology fit?
Use our spreadsheet to orient yourself, then go deep on the papers to build confidence. Don’t stop at a prominent academic or company saying that e.g. senescence is very important in disease X, get into the data. Ultimately drug programs succeed or fail on whether the drug you build is solving the right problem.
Are there compelling preclinical models, or will you have to take things into humans?
When animal models are a poor fit for human disease, as in pulmonary fibrosis or Alzheimer’s, investors will be less impressed with even strong preclinical data. This means you might have to raise enough money to run human trials before things really take off. Can you do that? Look at the key readouts for human trials, does your data suggest you can move those measures?
What will it take to run human trials?
Speaking of human trials, how long will they take? How much money will you burn during those years? And how much will the trials cost? As a rough guideline, assume $50K-200K per patient. If trials are slower and more expensive, you’ll need a more impressive drug to motivate investment.
Is there an unmet need?
How good is the standard of care treatment, and how would your drug differentiate? It’s not enough to have something that works, you need to be either better than or additive with what’s already on the market. Look at the drugs in clinical trials too (not included here). You might also consider whether you’ll be able to pivot to another indication if something really good gets approved while you’re building.
We hope this overview will be useful, but you’ll still have to think deeply about therapeutic strategy. Indication selection is at the intersection of business and science decisions, and having a crisp picture (while being willing to pivot as you learn) will help you go further on what you’ve raised so far.
This resource is free. It was built by Martin Borch Jensen and Longevity Apprentices Madi Ueland and Divya Cohen, with input from Norn Nexus members.
If you find an error or an omission, please reach out.
age1 wrote a nice overview of other key decisions:
Or if that innovation is your business model and you have a unique angle on it, like Loyal, they’ll want to see incredibly derisked science.




What struck me reading this is that the core challenge you describe – selecting a disease as an entry point for aging – may not just be a regulatory problem, but a conceptual one.
Aging is being approached as something that needs a proxy (a disease), while it might be more accurate to understand it as a gradual shift in regulatory processes across systems.
In that sense, the difficulty of “indication selection” could reflect a deeper mismatch: we are trying to anchor a dynamic, system-level process in static, disease-based endpoints.
If aging is fundamentally about changes in regulation – in how systems maintain stability under varying demands – then the question may not only be which indication to choose, but whether the indication framework itself is sufficient to capture what is actually changing.
And this leads to a second question:
If the underlying issue is a loss of regulatory capacity, is the primary need really a drug – or a shared literacy about how biological systems maintain stability in the first place?
Perhaps both will play a role. But without a clearer understanding of the regulatory processes themselves, even well-designed interventions risk being applied to the wrong level of the system.